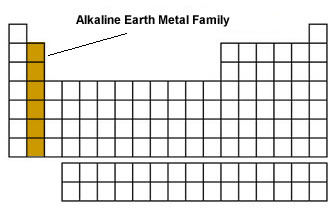
(1) The divalent cation of alkaline earth metal acquire stable inert gas configuration. 
Alkaline earth metals uniformly show an oxidation state of +2.Īlkaline earth metal prefer to form divalent ions rather than monovalent ions Therefore, they can easily lose these two electrons to form divalent cation. The alkaline earth metals have two electrons more than the nearest noble gas configuration. The compounds of alkaline earth metals are more extensively hydrated than those of alkali metals. Reason: Due to smaller size of alkaline earth metal ions as compared to alkali metal ions ,the hydration enthalpies of alkaline earth metal ions are larger than those of alkali metal ions. The hydration enthalpies of alkaline earth metal ions decreases as the size of the metal ion increases down the group i.e. The third ionization enthalpy of magnesium will be very high because now the electron has to be removed from the stable noble gas configuration. Therefore, removal of second electron in case of alkaline earth metals requires much less energy than that in case of alkali metals. Reason: The second electron in case of alkali metal is to be removed from a cation which has already acquired the stable noble gas configuration whereas in case of alkaline earth metal, the second electron is to be removed from a cation which is yet to acquire the stable noble gas configuration. The second ionization enthalpies of the elements of group 1 are higher than those of elements of group 2. On moving down the group, ionization enthalpy values go on decreasing because of the increase in atomic size due to addition of the new shells and screening effect of the electrons in the inner shell which overweigh the effect of increased nuclear charge. The values of the first ionization enthalpy of elements of Group 2 are greater than those of the elements of group 1 because the atoms of alkaline earth metals have smaller size and higher nuclear charge than those of alkali metals. Reason: The low ionisation enthalpies of the alkaline earth metals is because of their strong tendency to lose electrons due to their smaller nuclear charge and comparatively larger atomic size which results in weaker forces of attraction between the valence electrons and the nucleus. The alkaline earth metals have fairly low ionization enthalpies though greater than those of the corresponding elements of group 1 and these decreases down the group. On moving down the group, the atomic and ionic radii increases due to addition of an extra shell of electrons in each succeeding element and the increasing screening effect. As a result ,their atomic and ionic radii are smaller than those of the corresponding alkali metals. Reason: The alkaline earth metals have a higher nuclear charge ,and ,therefore the electrons are attracted more strongly towards the nucleus. 5 (6) Electropositive or Metallic CharacterĪtomic and ionic radii of alkaline earth metals are fairly large though smaller than the corresponding alkali metals and these increases down the group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
